Surgical Bandage Manufacturing Plant: Cost, Setup, Machinery & Business Plan

Why Surgical Bandage Manufacturing is a Profitable Opportunity
The demand for surgical bandages is consistently high due to their essential use in hospitals, clinics, and home healthcare. Unlike many medical products, bandages are consumables, meaning repeat demand is guaranteed.
With the expansion of healthcare infrastructure and increasing awareness of wound care, setting up a surgical bandage manufacturing plant presents a low-risk, high-volume business opportunity.
Market Demand & Growth Potential
- Surgical bandages are widely used in:
- Hospitals and clinics
- Emergency care
- Home healthcare
- Key growth drivers:
- Increase in surgeries and injuries
- Rising demand for first-aid products
- Government healthcare initiatives
This makes bandage manufacturing a stable and scalable business model.
Types of Surgical Bandages You Can Manufacture
1. Adhesive Bandages
- Common “band-aid” type products
- High demand in retail markets
2. Gauze Bandages
- Used for wound dressing
- Require sterilization
3. Elastic Bandages
- Used for orthopedic support
- Require elasticity and durability
4. Crepe Bandages
- Widely used for compression therapy
Step-by-Step Manufacturing Process
1. Raw Material Procurement
- Cotton gauze
- Medical-grade adhesive
- Fabric and elastic materials
Quality of raw materials directly impacts product safety and compliance.
2. Cutting & Shaping
- Fabric is cut into required sizes
- Automated cutting machines improve efficiency
3. Pad Formation (for adhesive bandages)
- Absorbent pad is placed at the center
- Ensures wound protection
4. Adhesive Coating
- Medical-grade adhesive is applied
- Must be skin-friendly and non-irritating
5. Rolling / FOLDING
- Bandages are rolled or folded
- Prepared for packaging
6. Sterilization
Common methods:
- Ethylene Oxide (EtO)
- Gamma radiation
Sterilization is critical for infection prevention.
7. Packaging
- Individual sterile packaging
- Bulk hospital packaging
8. Quality Control
- Adhesion testing
- Absorbency testing
- Sterility validation
Cost of Surgical Bandage Manufacturing Plant
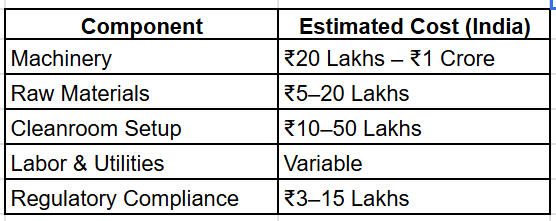
Total Investment: ₹50 Lakhs — ₹2+ Crore
Machinery Required
- Bandage cutting machine
- Adhesive coating machine
- Rolling / folding machine
- Sterilization equipment
- Packaging machine
Automation level directly impacts production capacity and cost.
Regulatory Requirements (VERY IMPORTANT)
To manufacture and sell surgical bandages, compliance is mandatory:
India
- Medical Device Rules (CDSCO)
- Manufacturing license
Global Standards
- ISO 13485 (Quality Management System)
- CE Marking (Europe)
- FDA registration (USA)
Non-compliance can lead to product rejection and legal issues.
Cleanroom & Sterility Requirements
- Controlled environment for sterile products
- HEPA filtration
- Environmental monitoring
Required especially for export-grade products.
Profit Margin & ROI
- High-volume product → consistent revenue
- Profit margins: 15%–30% (approx.)
- Break-even period: 1.5–3 years
Strong distribution network = higher profitability
Common Mistakes to Avoid
- Using low-quality raw materials
- Ignoring sterilization validation
- Lack of regulatory planning
- Poor packaging quality
These mistakes can damage brand reputation and lead to rejection.
How to Start a Surgical Bandage Manufacturing Business
- Conduct market research
- Prepare business plan
- Select plant location
- Procure machinery
- Obtain regulatory approvals
- Set up quality systems (ISO 13485)
- Launch production and distribution
Expert Support for Faster Setup
Setting up a compliant manufacturing plant requires technical, regulatory, and operational expertise.
At Operon Strategist, we support:
- End-to-end manufacturing setup
- Regulatory approvals (CDSCO, FDA, CE)
- ISO 13485 implementation
- Cleanroom design and validation
Learn more: https://operonstrategist.com/


.jpg)
Comments
Post a Comment