How to Start the Manufacturing of Disposable Syringes: A Practical Guide for New Manufacturers
The demand for disposable syringes has moved far beyond hospitals it is now driven by mass vaccination programs, chronic disease management, and the global shift toward single-use medical devices. For entrepreneurs and medical device manufacturers, this creates a strong business opportunity. However, entering this space requires more than basic setup it demands regulatory readiness, precision manufacturing, and validated processes.
Understanding the Regulatory Foundation (India Focus)
Before investing in machinery or infrastructure, manufacturers must align with regulatory requirements. In India, disposable syringes fall under notified medical devices and require approval from the Central Drugs Standard Control Organization (CDSCO).
To begin manufacturing:
- Obtain a CDSCO Manufacturing License (MD-5/MD-9)
- Implement a Quality Management System as per ISO 13485
- Ensure product classification and technical documentation are in place
Regulatory approval is not just a legal requirement it directly impacts your ability to supply to hospitals, participate in tenders, and export globally.
Facility Setup: Why Cleanroom Design Matters
Disposable syringes are sterile, single-use devices that come in direct contact with the human body. This makes controlled manufacturing environments essential.
- ISO-classified cleanrooms for molding and assembly
- Controlled air handling (HVAC) systems
- Segregated zones for raw materials, production, and packaging
Poor facility design often leads to contamination risks, batch rejection, and regulatory non-compliance one of the most common challenges faced by new manufacturers.
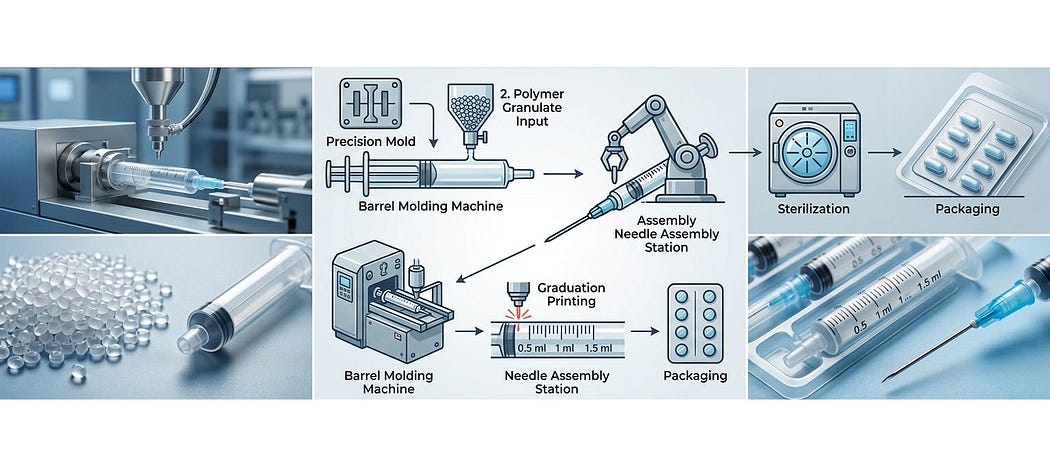
Core Manufacturing Process (What Actually Happens on the Floor)
The manufacturing of disposable syringes is a highly automated and precision-driven process:
- Injection Molding:
Medical-grade polypropylene is molded into syringe components (barrel, plunger) using high-speed injection molding machines. - Component Assembly:
Automated assembly lines integrate barrels, plungers, gaskets, and needles with minimal human intervention. - Siliconization & Lubrication:
Internal surfaces are treated to ensure smooth plunger movement and accurate dosing. - Sterilization (EO Process):
Ethylene Oxide (EO) sterilization is used to ensure the product is free from microbial contamination. - Blister Packaging:
Sterile syringes are packed in tamper-proof packaging for safe distribution.
Each of these steps must be validated and documented, especially sterilization, which is critical for patient safety.
Machinery & Investment Considerations
Setting up a syringe manufacturing unit involves capital-intensive equipment such as:
- Multi-cavity injection molding machines
- Fully automated assembly lines
- EO sterilization chambers
- Blister packaging systems
The actual investment depends on production scale and automation level. Manufacturers targeting export markets must also invest in advanced quality control systems and validation protocols.
Raw Materials That Define Product Quality
The quality of a syringe is not just about design — it heavily depends on material selection. Key materials include:
- Polypropylene (PP): For barrel and plunger
- Elastomer (Rubber): For gasket sealing
- Stainless Steel: For needles
- Silicon Oil: For lubrication
Using non-certified or low-grade materials can result in product failure during testing or rejection during audits.
Market Opportunity: Why This Industry Is Growing Fast
The disposable syringe market continues to expand due to:
- Increased vaccination drives and public health programs
- Rising prevalence of chronic diseases (diabetes, autoimmune conditions)
- Growing adoption of self-injection devices
- Strict regulations against reuse of needles
Manufacturers who align with quality and compliance standards can tap into both domestic and export markets.
Common Challenges New Manufacturers Face
Many first-time manufacturers underestimate the complexity of this industry. The most common challenges include:
- Delays in CDSCO approvals due to incomplete documentation
- Difficulty in achieving consistent product quality at scale
- Improper EO sterilization validation
- Poor cleanroom design leading to contamination
These challenges often lead to increased costs, delayed market entry, and product rejection.
The Role of Expert Guidance in Faster Market Entry
Given the regulatory and technical complexity, many manufacturers partner with experienced consultants to:
- Design compliant manufacturing facilities
- Prepare regulatory documentation
- Implement ISO 13485 systems
- Validate sterilization and production processes
This significantly reduces risk and accelerates time-to-market.
Conclusion
Starting a disposable syringe manufacturing business is not just about capital investment it is about building a compliant, quality-driven production system from day one. Manufacturers who focus on regulatory alignment, validated processes, and cleanroom-controlled production can achieve faster approvals and long-term scalability in a highly competitive market.


.jpg)
Comments
Post a Comment